According to this theory, the electron transfer by the respiratory chain complexes leads to vectorial extrusion of protons from the matrix into the intermembrane space of mitochondria. 
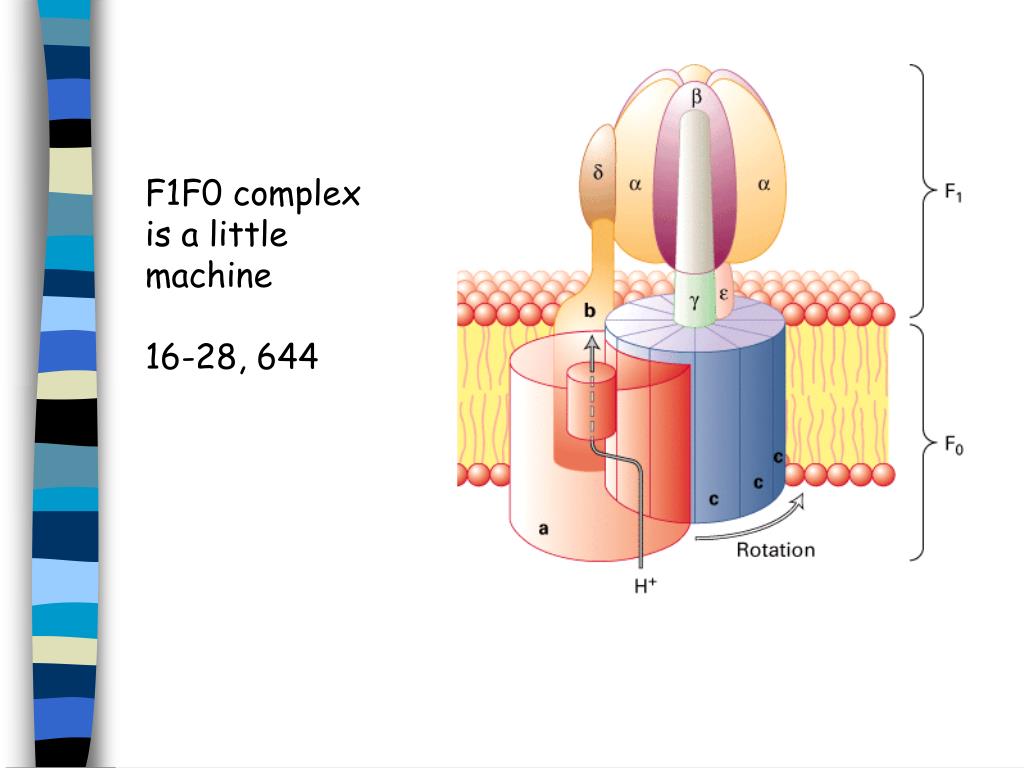
The mechanism coupling respiration to ATP synthesis remained enigmatic, however, until Peter Mitchell formulated the chemiosmotic theory in 1961. In 1925 Otto Warburg first described the phenomenon of cellular respiration, , and 15 years later Herman Kalckar showed that respiration was linked to ATP production. In this scheme the structural models are not drawn to scale. Schematic depiction of the oxidative phosphorylation system. In this Primer, we will briefly introduce the recent progress made in this area and highlight the road ahead that likely will unravel the detailed molecular mechanisms of complex I function.įigure 1. Low-resolution X-ray structures of complex I have, surprisingly, suggested that electron transfer in the hydrophilic arm, protruding into the mitochondrial matrix, causes movement of a coupling rod that influences three putative proton pumps within the hydrophobic arm embedded in the inner mitochondrial membrane. The molecular mechanism whereby it couples electron transfer to proton extrusion has remained mysterious until very recently. Complex I (or NADH-quinone oxidoreductase) is the largest and by far the most complicated of the respiratory chain enzyme complexes. This process involves a series of large enzyme complexes-the respiratory chain-that couples the transfer of electrons to the creation of a concentration gradient of protons across the inner mitochondrial membrane, which drives ATP synthesis. Mitochondria are the structures that produce the bulk part of the cellular energy currency ATP, which drives numerous energy requiring processes in the cell. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
